Infusion of Furosemide to Improve Diuretic Efficiency in Acute Heart Failure (INFUSE-AHF)

Title
Infusion of Furosemide to Improve Diuretic Efficiency in Acute Heart Failure (INFUSE-AHF)
Aim
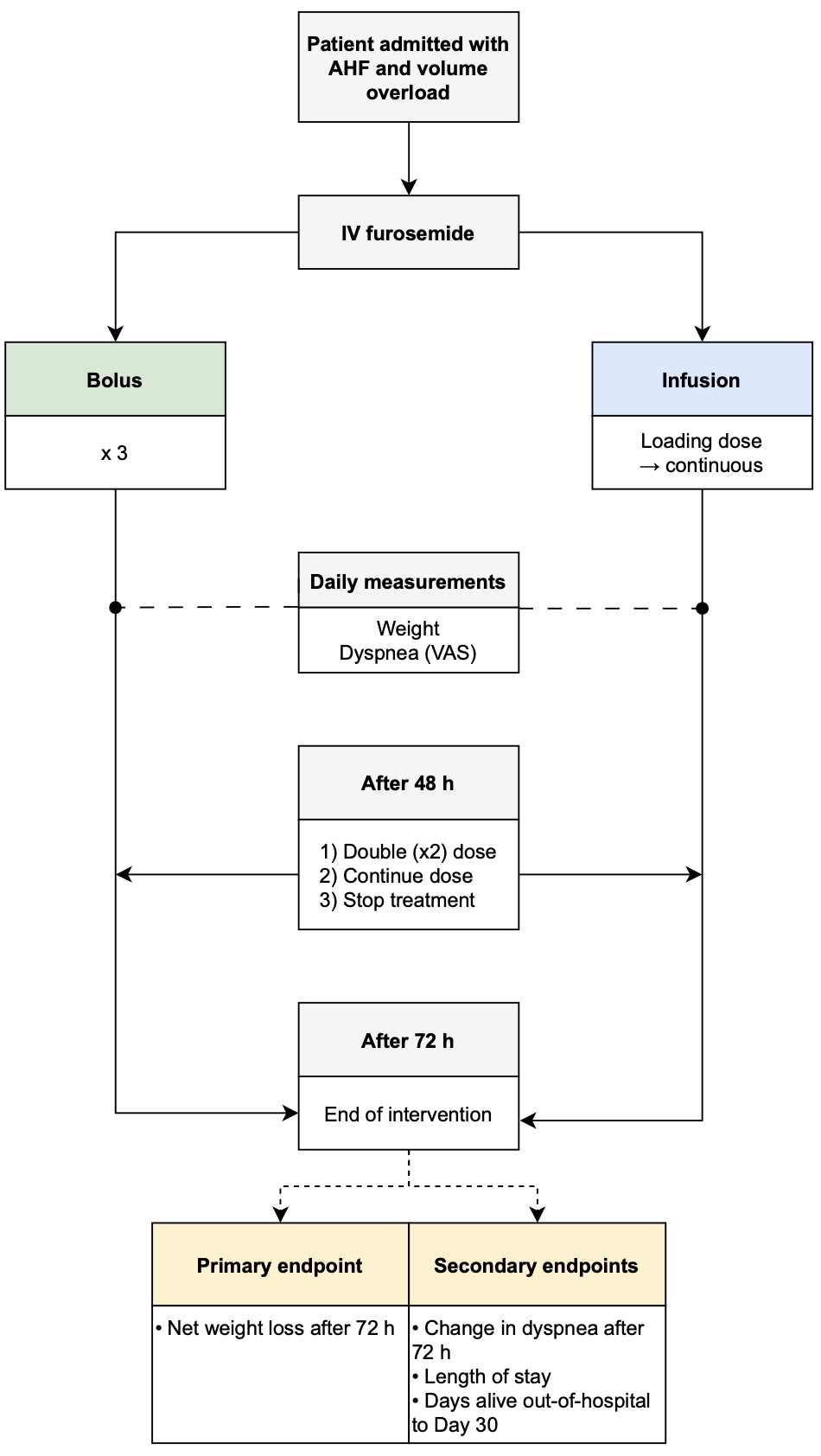
To determine if there is a clinically meaningful difference in the diuretic treatment effect in acute heart failure between intravenous administration of furosemide by either continuous infusion preceded by a loading dose or bolus injections three times a day.
Design
Investigator-initiated, multicenter, open-label, pragmatic, randomized clinical trial.
Methods
Adult patients admitted with acute heart failure and ≥1 sign of volume overload are randomized 1:1 to intravenous furosemide by continuous infusion preceded by a loading dose or bolus injections three times daily.
Population
Approximately 436 patients with acute heart failure and volume overload from Emergency and Cardiology Departments in Denmark.
Outcome
Primary endpoint:
- Net weight loss 3 days (≈72 hours) after randomization
Secondary endpoints:
- Net weight loss 2 days (≈48 hours) after randomization
- Change in dyspnea on a Numeric Rating Scale 3 days (≈72 hours) after randomization
- Days alive out-of-hospital to Day 30
- Length of stay
Exploratory endpoints:
- Net weight loss 1 day (≈24 hours) after randomization
- Change in dyspnea on a Numeric Rating Scale 1 day (≈24 hours) after randomization
- Change in dyspnea on a Numeric Rating Scale 2 days (≈48 hours) after randomization
- Death from any cause after 30 days
- Rehospitalization from any cause after 30 days
Safety endpoints:
- Acute kidney injury
- Electrolyte disturbances
- Incidents and side effects
Perspectives
Loop diuretics are first-line therapy for acute heart failure, but robust evidence on optimal dosing and mode of administration is lacking. By testing a guideline-recommended but unproven strategy (continuous infusion with loading dose) against current standard bolus therapy in a real-world Danish setting, INFUSE-AHF may improve evidence-based diuretic treatment and inform future guidelines.
Steering Committee
Coordinating investigator: Esben Merrild, MD, PhD student, Department of Medicine (Cardiology), Randers Regional Hospital
Sponsor-investigator: Bo Løfgren, MD, PhD, FAHA, FESC, Department of Medicine (Cardiology), Randers Regional Hospital
Henrik Birn, PhD, DMSc, Professor, Department of Renal Medicine, Aarhus University Hospital
Kasper Glerup Lauridsen, MD, PhD, Associate Professor, Department of Medicine (Cardiology), Randers Regional Hospital
Christian Bo Poulsen, MD, PhD
Funding
Health Research Foundation of Central Denmark Region
Randers Regional Hospital
Department of Clinical Medicine, Aarhus University
Grosserer L. F. Foghts Fond
Registrations number
EU CT number: 2025-523589-26-00
Contact